Fiberoptic Endoscopic Examination of Swallowing in Laryngomalacia Patients
Article information
Trans Abstract
Background and Objectives
Laryngomalacia is the most common cause of stridor in infants and patients frequently present with feeding and respiratory complications. Fiberoptic Endoscopic Evaluation of Swallowing (FEES) is essential for diagnosing swallowing dysfunction in this population. This study describes the FEES findings and clinical characteristics of pediatric laryngomalacia patients in a tertiary referral setting.
Subjects and Method
This retrospective study included descriptive analyses of 21 laryngomalacia patients who underwent FEES between January 2021 and December 2024. Clinical variables included laryngomalacia type and severity, comorbidities, symptoms, and FEES outcomes. Food consistencies tested were adjusted by age and swallowing ability.
Results
Most patients were male (71.5%) with type I (80.9%) and severe-grade laryngomalacia (71.4%). Comorbidities were present in 76.2%, and swallowing symptoms in 95.2%. FEES identified 14 swallowing abnormalities across consistencies, with penetration (42.8%) and residue (28.5%) being most common. Thick liquids (milk) accounted for the majority of abnormalities. Patients with tracheostomy or severe laryngomalacia demonstrated a higher frequency of dysfunction. Silent aspiration was observed in 14.2% of patients.
Conclusion
Swallowing dysfunction is prevalent in infants with laryngomalacia, particularly in those with comorbidities and severe anatomical involvement. FEES is a valuable tool for early identification of penetration, aspiration risk, and silent dysphagia, enabling targeted management in high-risk populations.
Introduction
Laryngomalacia is a congenital abnormality of the larynx responsible for 70% of cases of stridor in infants [1]. Stridor is a high-pitched breathing sound that indicates an obstruction in the airway, particularly in the larynx and/or tracheobronchial region. Stridor generally worsens when the patient cries, feeds, or is in a supine position. Patients with laryngomalacia typically experience inspiratory stridor during the first few weeks of life, which often worsens and peaks in severity at 6 months of age, followed by gradual improvement of symptoms, with some patients becoming completely symptom-free by 18-24 months of age [2]. Although generally self-limiting and capable of resolving on its own, 10%-20% of laryngomalacia cases require surgical intervention [1-3].
Laryngomalacia can disrupt the coordination between suction/swallowing and respiratory functions, with patients also experiencing difficulties in feeding, failure to thrive, dysphagia, aspiration, obstructive sleep apnea (OSA), and even pulmonary hypertension in severe cases [1,2]. The actual incidence of dysphagia and aspiration in infants with laryngomalacia is unknown. Previous studies have indicated that infants with more severe laryngomalacia tend to exhibit symptoms of feeding difficulties [1]. Furthermore, there is evidence that infants with comorbid conditions such as neurological disorders or congenital heart disease tend to have a more severe form of laryngomalacia [3]. In diagnosing and evaluating the complications, a thorough clinical history and physical examination, combined with additional diagnostic tests such as Fiberoptic Endoscopic Evaluations of Swallowing (FEES), are necessary to assess swallowing function [2,3].
FEES examination is important for patients with laryngomalacia, particularly those experiencing swallowing difficulties. This procedure is performed by an ENT specialist and can be carried out at the bedside. It allows for direct anatomical assessment and evaluation of the sensory function of the pharynx, supraglottis, and glottis [4]. Thus, anatomical variations and malformations causing dysphagia can be easily detected [5,6]. This procedure can also be performed on infants who are breastfeeding and even on children who do not eat orally by observing the swallowing of colored saliva. The procedure is comfortable, safe, and provides meaningful diagnostic information [3,4,7].
This study aims to characterize FEES findings in infants with laryngomalacia at a tertiary referral hospital, focusing on clinical features, comorbidities, and swallowing abnormalities, contributing to better diagnosis and care..
Subjects and Methods
Study design
A retrospective descriptive study was conducted using medical records of laryngomalacia patients who underwent FEES between January 2021 and December 2024 at our hospital’s Department of Otorhinolaryngology-Head and Neck Surgery (ENT-HNS). This study includes a detailed description of the demographic characteristics (age, gender) and clinical conditions of laryngomalacia (type, degree, respiratory symptoms, swallowing symptoms, dysphagia, complications, silent aspiration, comorbidities, tracheostomy, and history of intubation). The research is conducted at the ENT-HNS at our hospital. The study is carried out in 2025, with the research process involving data collection and the preparation of the final report.
This study was approved by the Research Ethics Committee of RSUP Dr. Mohammad Hoesin Palembang (No. DP.04.03/D.XVIII.06.08/ETIK/078/2025). The potential participants were informed that their responses to the questionnaire would indicate consent for their participation in the study.
Population and sample
The sample in this study includes all laryngomalacia patients who underwent FEES at our hospital between January 2021 and December 2024 and whose medical records were complete. The sample size in this study comprises the entire accessible population that meets the inclusion criteria. Total sampling was applied.
FEES procedure
The FEES was performed by experienced ENT specialists using a flexible fiberoptic endoscope. Infants were examined while awake, either in a caregiver’s lap in upright position or semi-reclined. Food consistencies tested included thick liquids (milk), strained porridge, and oatmeal, as tolerated. Infants who were non-oral feeders were evaluated using colored saliva techniques. Safety was prioritized, and no sedation was used. Each examination was recorded and later reviewed by two raters to ensure reliability.
Reliability measure
To ensure intra- and inter-rater reliability, dual assessment was conducted. Discrepancies were resolved by consensus, enhancing validity of FEES interpretation.
Data collection and analysis
The data collected in this study consists of secondary data derived from the medical records of laryngomalacia patients who underwent FEES at the ENT-HNS at our hospital. Data were coded and analyzed using SPSS ver 25.0 (IBM Corp.). Univariate descriptive analysis summarized frequencies and distributions of clinical features and FEES outcomes.
Results
A total of 21 laryngomalacia patients who underwent FEES were included. The mean age was 10.4 months (range: 2-24 months), and the majority were male (71.5%). Most patients had type I laryngomalacia (80.9%), with severe cases comprising 71.4%. Notably, 57.1% also had type II laryngomalacia, indicating that several patients presented with mixed types. Only 14.3% of patients had mild or moderate forms, while the majority were classified as severe.
Comorbidities were found in 76.2% of patients, including neurological, cardiac, and genetic conditions. Swallowing symptoms were present in 95.2%, and respiratory symptoms were reported in 52.4%. Dysphagia was noted in 57.1% of patients, while silent aspiration was observed in 14.2%. Complications were present in 71.5%, and 33.3% of the cohort had undergone tracheostomy. The same percentage had a history of intubation.
FEES findings
Across all patients, 14 abnormal swallowing events were documented during the FEES procedure. The most frequent finding was penetration (6 cases), followed by residue (4 cases), leakage (3 cases), and aspiration (1 case). The majority of abnormalities were noted with thick liquid (milk), observed in 11 of 14 abnormal findings. Rice porridge accounted for 3 findings (penetration and residue), while other consistencies such as oatmeal, wheat porridge, thin liquids, and biscuits/crackers yielded no abnormalities.
Patients with tracheostomy (n=7) demonstrated a higher proportion of abnormal FEES findings, particularly penetration and residue. Similarly, patients with severe laryngomalacia and those with comorbidities were more likely to exhibit dysphagia and silent aspiration.
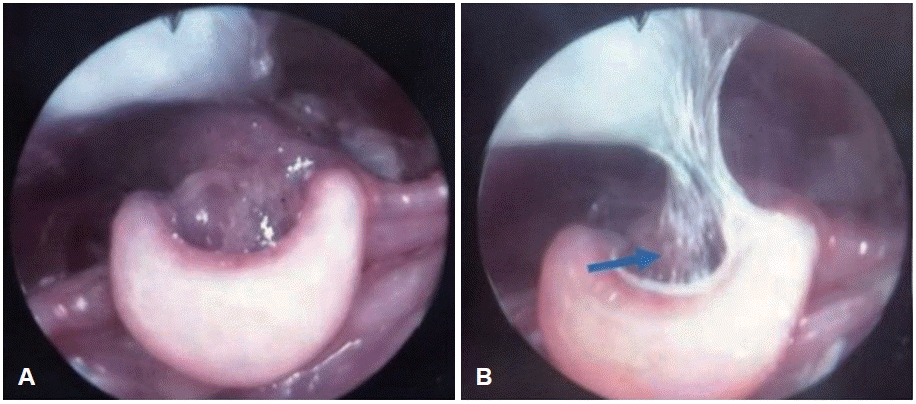
Representative endoscopic images obtained during FEES are provided in the Figs. 1 and 2. These include examples of penetration with thick liquids and pharyngeal residue, demonstrating typical findings in severe laryngomalacia cases. The images support the observed trends and provide visual context for interpreting the swallowing abnormalities described.
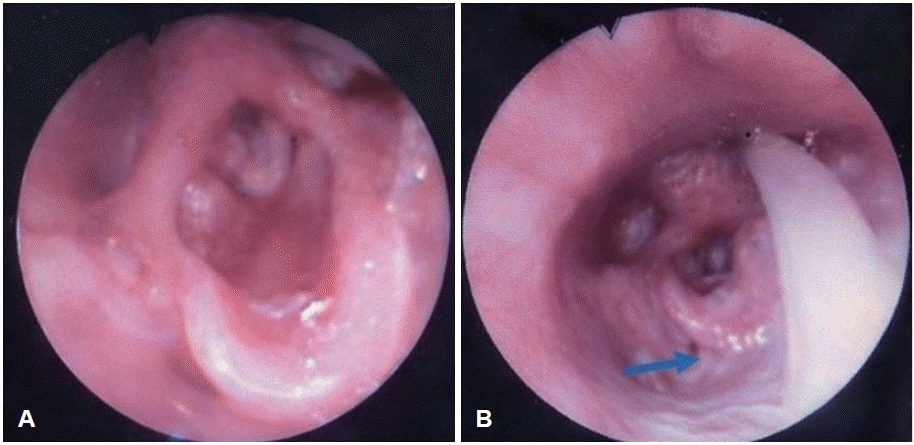
FEES findings on case 1. Patient using nasogastric tube. A: An omega shaped epiglotis. B: Residue in vallecula (blue arrow). FEES, Fiberoptic Endoscopic Evaluation of Swallowing.
Discussion
This retrospective study of 21 pediatric laryngomalacia cases provides detailed insights into FEES findings and clinical characteristics. Consistent with previous reports, most patients had type I laryngomalacia and were male. Severe cases dominated our sample, which likely reflects tertiary-level referral bias, as milder cases often do not undergo endoscopic evaluation.
A noteworthy proportion (57.1%) showed overlapping features of type I and II, reinforcing the concept that laryngomalacia can present as a spectrum. Our findings align with Pokhare [l8] and Bredun, et al. [9], who also reported mixed-type presentations.
The high incidence of comorbidities (76.2%) is consistent with literature showing associations between laryngomalacia and conditions such as Down syndrome, GERD, and neurologic disorders [10-12]. These comorbidities contribute to symptom severity and swallowing dysfunction. However, our study lacked sufficient power to analyze statistical correlations between comorbidity subtypes and FEES outcomes.
In this study, 52.4% of patients with laryngomalacia exhibited respiratory symptoms, with inspiratory stridor being the most common, exacerbated by eating, agitation, supine position, and crying. Stridor often improves or disappears during sleep in mild to moderate cases. Respiratory issues such as OSA, tachypnea, dyspnea, respiratory distress, or hypoxemia occurred in 5%-20% of children, and in severe cases, pulmonary hypertension and cor pulmonale may develop without proper treatment [13]. While infants with stridor and no significant feeding-related symptoms can be monitored without surgery, supraglottic airway obstruction from laryngomalacia can increase respiratory effort and energy demands, potentially leading to poor weight gain or even muscle atrophy when combined with feeding difficulties [3,14].
The study also found that 95.25% of patients experienced swallowing issues, with 49% showing symptoms like regurgitation or aspiration, though no clear relationship between these conditions has been established. Previous studies suggest that swallowing problems in laryngomalacia patients may be linked to comorbid conditions such as neurological disorders or respiratory reflux diseases [2,12]. Silent aspiration, where food or liquid enters the airway without triggering a cough reflex, was observed in three patients, which is common in around 40% of swallowing dysfunction cases and can lead to serious respiratory conditions like aspiration pneumonia. The study also found that 57.14% of cases had dysphagia, with similar findings in other studies, although the prevalence of swallowing difficulties is often underestimated due to silent aspiration. The relationship between laryngomalacia and dysphagia is not well-understood but may involve mechanical obstruction during swallowing, increased subglottic negative pressure, and disturbances in the suck-swallow-breath sequence, as well as impaired laryngeal protection reflexes and reduced supraglottic mucosal sensitivity. Gastroesophageal reflux further exacerbates dysphagia by damaging the mucosa and decreasing laryngeal reflex sensitivity [13,15].
Penetration and residue were the most common findings on FEES. These abnormalities were predominantly observed with thick liquid (milk), highlighting the importance of evaluating consistency-specific swallowing function in infants. The solitary aspiration event observed also occurred with thick liquids. These results are consistent with Ha [1] and Simons, et al. [2], who also found thick liquids to be the most problematic consistency in infants with laryngomalacia. Richter and Thompson [16] reported 88% penetration and 72% aspiration in severe laryngomalacia patients who underwent supraglottoplasty, with 82% of patients with penetration and 86% with aspiration experiencing resolution post-surgery.
Laryngeal penetration occurs when food enters the larynx but does not pass the true vocal cords, and when deep laryngeal penetration occurs, 85% lead to aspiration. Residue is associated with oral phase changes and delayed swallowing reflex initiation. Clinical swallowing assessments should be conducted on all patients with laryngomalacia, even without subjective dysphagia symptoms [4,12,15]. These findings highlight the importance of conducting swallowing function assessments for all laryngomalacia patients, regardless of whether they show dysphagia symptoms.
Importantly, patients with tracheostomy and severe grades exhibited a higher rate of penetration and residue. Although formal statistical testing was not feasible due to the small sample, this trend suggests that FEES can aid in identifying high-risk subgroups who may benefit from modified feeding strategies or further intervention.
The presence of silent aspiration in 14.2% supports earlier recommendations for routine instrumental swallowing assessments, even in patients who lack overt symptoms. Our data confirms the diagnostic value of FEES in uncovering clinically silent but significant swallowing issues.
Intra- and inter-rater reliability were maintained through double reading and consensus discussions, helping to mitigate subjectivity.
Limitations include the retrospective nature of the study, small sample size, and the absence of standardized scoring for FEES. Nevertheless, the results underscore the need for early swallowing assessment in laryngomalacia patients, especially those with comorbidities or respiratory compromise.
In summary, this study reinforces that FEES is an essential tool in the multidisciplinary management of pediatric laryngomalacia. Future research should include larger, prospective cohorts with structured FEES scoring and detailed comorbidity analysis.
In conclusion, this study highlights that swallowing dysfunction is common in pediatric patients with laryngomalacia, particularly those with severe disease, comorbidities, or a history of tracheostomy. Penetration and residue were the most frequent abnormalities observed on FEES, predominantly with thick liquid consistencies such as milk. Silent aspiration was also present in a subset of patients, reinforcing the need for instrumental evaluation even in the absence of overt symptoms.
Our findings support the routine use of FEES as a safe and effective diagnostic tool in evaluating feeding and swallowing in infants with laryngomalacia. It enables early detection of aspiration risks and guides personalized management, especially in high-risk subgroups. Despite the limitations of retrospective design and small sample size, the study underscores the clinical relevance of FEES in the multidisciplinary care of this population. Future prospective studies with standardized scoring systems are warranted to further validate the role of FEES and establish clear protocols for early intervention in laryngomalacia-related dysphagia.
Notes
Acknowledgments
Special thanks to medical staff at our hospital and institution for providing the data and facilitating the study.
Author Contribution
Conceptualization: Puspa Zuleika. Data curation: Puspa Zuleika, Rohayu. Formal analysis: Puspa Zuleika. Investigation: Puspa Zuleika. Methodology: Puspa Zuleika. Project administration: Puspa Zuleika. Supervision: Rohayu. Writing—original draft: Puspa Zuleika. Writing—review & editing: Puspa Zuleika, Rohayu.