Prevalence of Human Papillomavirus in Head and Neck Cancers in South Korea: A Targeted Literature Review
Article information
Trans Abstract
This study assessed the human papillomavirus (HPV) prevalence and genotypes in head and neck cancers in South Korea. We performed a targeted literature review of studies published between March 2002 and January 2023 reporting on HPV genotypes in head and neck squamous cell carcinomas (HNSCCs), including oral cavity, oropharynx, larynx (glottis), hypopharynx, tonsil, tongue, gum, posterior wall, and sinonasal tract in South Korea. Nineteen eligible studies were identified. The mean prevalence of any HPV genotype was 56.5% (range, 14.5%-75.1%) for oropharyngeal cancer and 7.5% (range, 3.0%-13.8%) for oral cavity cancer. The mean prevalence of any HPV genotype was higher in male (37.9%) than female (24.1%) cases. HPV-16 was the most common genotype detected in HPV-positive cancers, with a prevalence of 75.6% (range, 29.0%-100%) in oropharyngeal cancer, 74.1% (range, 29.0%-100%) in tonsil cancers, and 29.5% (range, 0.9%-84.6%) in tongue cancers. HPV-18 was detected in 9.0% (range, 0.0%-29.0%) of HPV-positive oropharyngeal cancers and 11.8% (range, 0.0%- 72.7%) of HPV-positive oral cavity cancers. In South Korea, HPV infection is an important factor in the development of HNSCCs, particularly oropharyngeal cancers, where HPV-16 was the predominant genotype. The high prevalence of HPV-16 in cancers of the oropharynx, tonsils, and tongue underscores its strong association with these malignancies. Additionally, HPV- 18, HPV-33, and other high-risk genotypes contribute to a smaller proportion of cases. These findings highlight the potential impact of HPV vaccination in reducing the burden of HPV-associated head and neck cancers in South Korea.
Introduction
Head and neck squamous cell carcinoma (HNSCC) includes lip-oral cavity cancer, laryngeal cancer, nasopharyngeal cancer, and other pharyngeal cancers. Worldwide, an estimated 19.96 million new cases of cancer were recorded in 2022, with 2.0% being lip-oral cavity cancer, 0.9% laryngeal cancer, and 0.6% nasopharyngeal cancer [1]. In the same year, there were 9.7 million cancer-related deaths globally, with 1.9% attributable to lip-oral cancer, 1.1% to laryngeal cancer, and 0.8% to nasopharyngeal cancer [1]. The incidence of HNSCCs is rising in multiple countries, including some countries in Asia [2]. In South Korea, HNSCCs constitute a substantial health burden and accounted for over 7000 new cancer diagnoses and over 3000 deaths in 2021 [3]. Recent studies indicate that human papillomavirus (HPV) is an important cause of HNSCCs globally, causing approximately 20%-70% of cancers of the oropharynx and about 7.4% and 5.7% of cancers of the oral cavity and larynx, respectively [4-8]. The proportion of HNSCCs due to HPV, though varying widely by location, is highest in more developed countries, including South Korea [5].
Oral HPV infection is now recognized as a key driver of global increases in cancers of the oropharynx, which are most pronounced among males in North America, Europe, and parts of Asia [9]. In South Korea, the incidence of cancer of the oropharynx has increased by over 2% annually since 2000, with the sharpest rise observed in males [10]. Furthermore, from 2008 to 2020, HPV infection accounted for a growing proportion of cancers of the oropharynx in South Korea [11], a trend similar to those observed in the United States and France [12,13]. This rising trend may place a substantial financial burden on South Korea’s healthcare system, in which the median treatment cost for cancer of the oropharynx was over $20000 (USD) in 2016 [14].
HPV vaccination, especially before sexual debut, may help prevent HPV-related cancers. Currently, 3 HPV vaccines are available in South Korea: a 2-valent vaccine (Cervarix™, GlaxoSmithKline), a 4-valent vaccine (Gardasil®, Merck & Co., Inc.), and a 9-valent vaccine (Gardasil®9, Merck & Co., Inc.) [15]. HPV vaccination in 12-year-old girls was introduced in South Korea’s National Immunization Program in 2016 [14,15]. However, routine HPV vaccination is not currently provided for boys [16,17]. Globally, greater consideration is being given to gender-neutral HPV vaccination, which has already been implemented in 33 of 38 member countries of the Organisation for Economic Co-operation and Development [16,18].
To help inform HPV vaccination policy, consolidated evidence regarding HPV-associated HNSCCs is essential but scarce for South Korea. Notably, current estimates regarding HPV-positivity in oral cavity cancers are based on a few case series, primarily from Europe and North America [5]. Moreover, to our knowledge, HPV positivity in HNSCCs in South Korea has been evaluated in only a few systematic reviews that included only some of the available studies in this area. To address this knowledge gap, the current study used data from HPV genotyping analyses in South Korean patients to comprehensively evaluate the prevalence and genotype distribution of HPV in HNSCCs by anatomical site.
Methods
A targeted review of the literature was conducted to identify studies published from March 2002 through January 2023 that reported prevalences of HPV genotypes among patients with HNSCCs in South Korea. This review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses 2020 reporting guidelines [19].
Search strategy
In June 2023 we conducted systematic searches in PubMed, Embase, and the Google Scholar database. Key search terms included “human papillomavirus,” “HPV,” “head and neck neoplasms,” “head and neck carcinoma,” “oropharynx,” “oropharyngeal,” “oral cavity,” “tonsil,” “tonsillar,” “tongue,” “hypopharyngeal,” “hypopharynx,” “nasal,” and “nasopharyngeal.” The search was limited to studies with the terms “Korea,” “South Korea,” “Seoul,” or “Republic of Korea.” A complete list of search terms and limits is provided in Supplementary Table 1.
Study inclusion and exclusion criteria
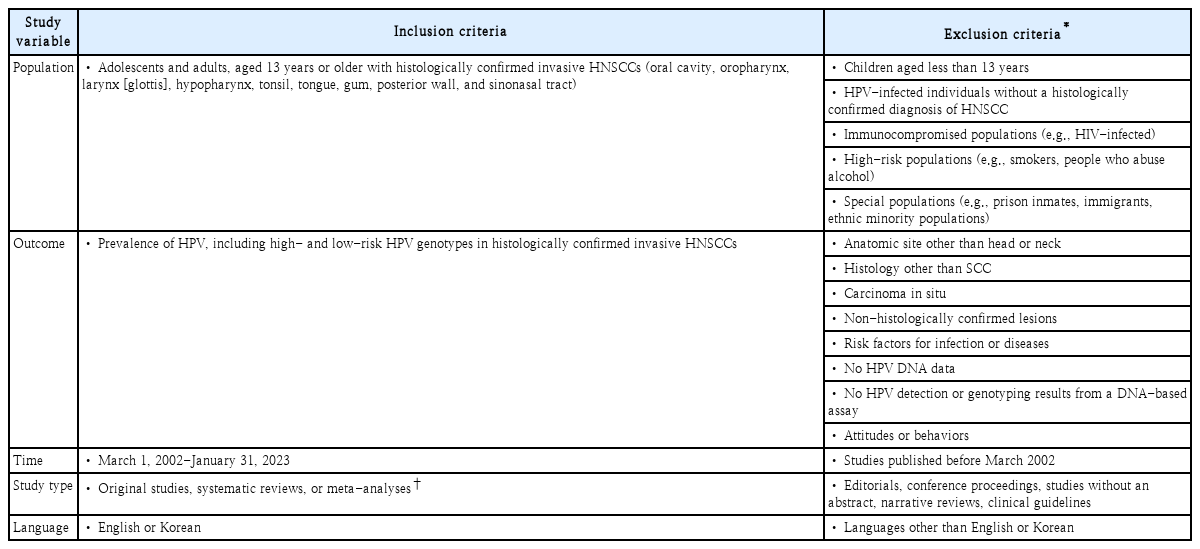
Studies included in this review were selected based on predefined criteria, as described in Table 1. Specifically, this review included original studies, systematic reviews, or meta-analyses reporting on the prevalence of HPV in adolescents and adults aged 13 years or older, with histologically confirmed invasive HNSCC. Outcomes of interest included the prevalence of high-risk (HR) HPV genotypes (16, 18, 31, 33, 35, 39, 45, 51, 52, 56, 58, and 59) and low-risk (LR) HPV genotypes [20]. Studies varied regarding the number of genotypes included in laboratory assays as well as genotype-based definitions of HR and LR HPV. Systematic reviews and meta-analyses were retrieved, and their reference lists were screened for potentially relevant articles. Studies reporting only on HPV prevalence in healthy populations or individuals with carcinoma in situ or malignancies in other anatomic sites were excluded.
Study selection
To identify potentially relevant studies, a single reviewer (AFSM) conducted an initial screen of titles and abstracts. Studies undergoing the initial screen were retained for full-text review if they fit or could not be excluded from fitting the inclusion and exclusion criteria based on the information in the title and abstract. Full-text versions of studies passing the initial screen were then evaluated by a single reviewer (AFSM), who conducted a detailed assessment to determine whether the study fit the inclusion and exclusion criteria. Reasons for exclusion were documented.
Data extraction and synthesis
Using a preset template, information on key parameters was extracted from eligible studies and entered into a Microsoft Excel workbook (Microsoft Version 16.86). Parameters included study characteristics (study name, first author, publication date, and study design), patient characteristics (overall sample size, the number of study subjects with HNSCC, HPV-positive HNSCC, HR HPV-positive HNSCC, LR HPV-positive HNSCC, and cancer type [see Supplementary Table 2 for cancer type classifications]) method of tissue collection, method of HPV DNA detection, and type of HPV genotyping test. The following outcomes were extracted: overall HPV prevalence in HNSCC tumors, the distribution of HR and LR genotypes in HPV-positive HNSCC tumors, the age distribution of patients with HPV-positive HNSCC tumors, and the sex distribution of patients with HPV-positive HNSCC tumors.
Descriptive statistics, including frequency distributions and percentages, were used to analyze the prevalence of any HPV and HR genotypes by anatomical site, age, and sex. The pooled sample size and prevalence range for any HPV, HR HPV, and HPV genotypes were reported by HNSCC subtype. Weighted mean prevalence values were calculated using weights based on the study sample size. Data were systematically tabulated and visualized using bar charts.
Results
Summary of study characteristics
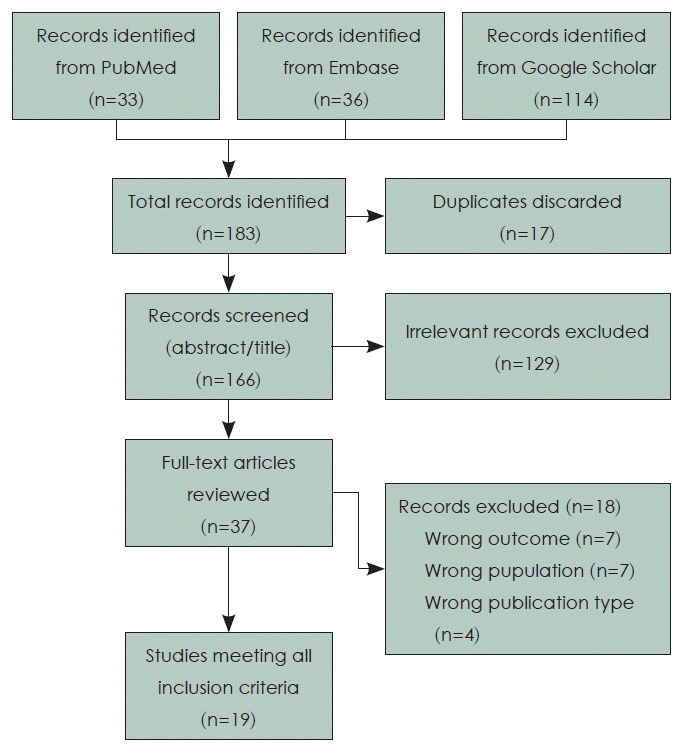
In total, 183 potentially eligible studies were identified, including 33 from PubMed, 36 from Embase, and 114 from Google Scholar (Fig. 1). After removing 17 duplicate records, 166 studies underwent screening of the title and abstract, of which 129 were excluded for not fitting the inclusion criteria. Thus, 37 studies underwent a full-text review, among which 18 were excluded for the following reasons: wrong outcome (n=7), wrong study population (n=7), and wrong study type (n=4).
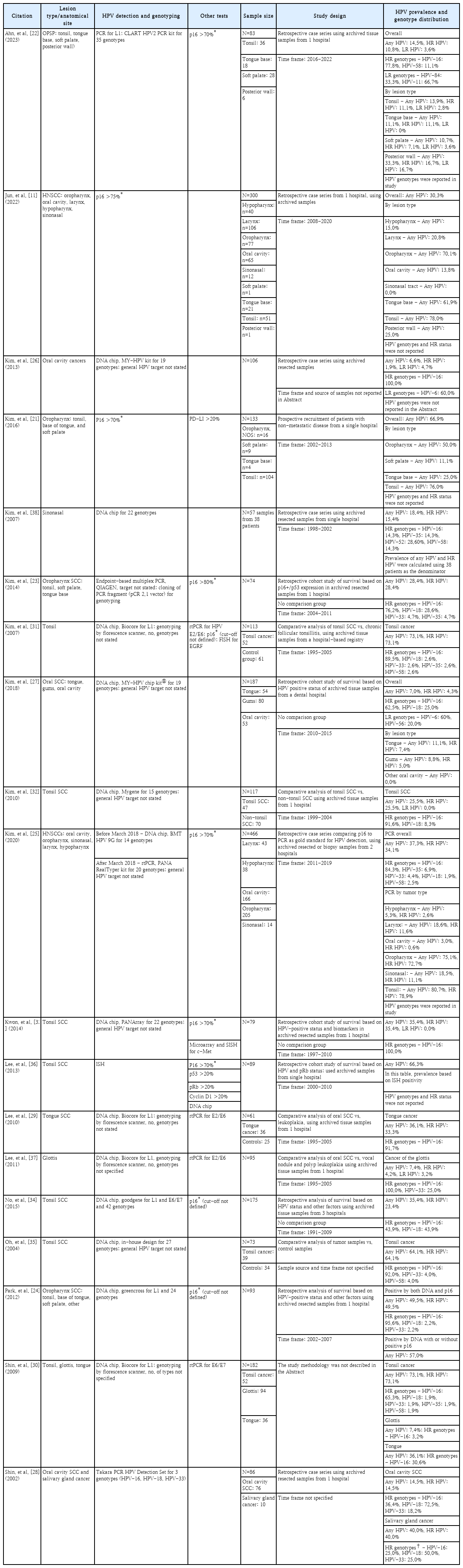
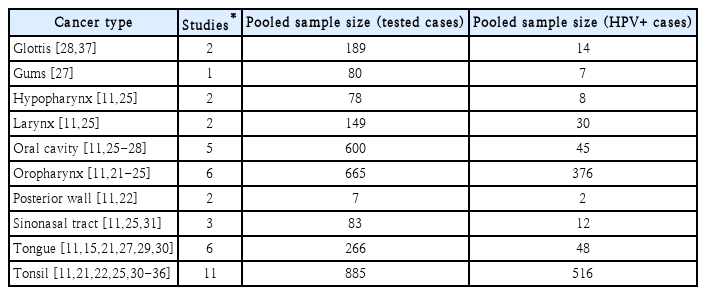
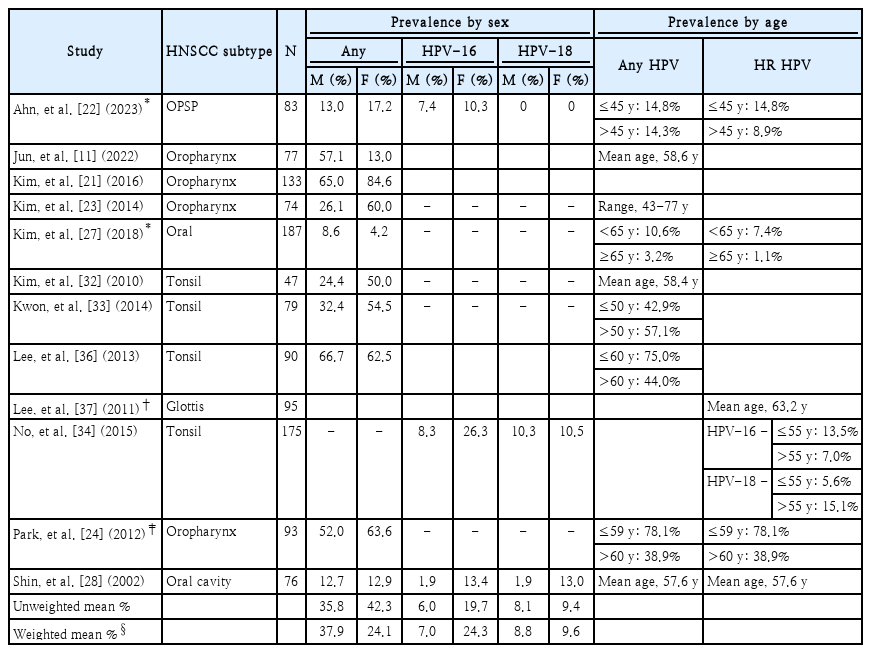
In total, 19 studies met all inclusion and exclusion criteria (Table 2); all but 1 study [21] were retrospective analyses. HNSCC evaluated in the studies included cancers of the oropharynx [11,21-25], oral cavity [11,25-28], tongue [11,15,21,27,29,30], tonsil [11,21,22,25,30-36], and glottis [28,37] (Table 3). Some studies evaluated multiple subtypes of HNSCC.11,25,30) The total pooled sample size of cancer cases evaluated was 3002, including 885 cancers of the tonsil from 11 studies, 665 cancers of the oropharynx from 6 studies, 600 oral cavity cancers from 5 studies, and 266 tongue cancers from 6 studies. In total, 12 studies [11,21-24,27,28,32-34,36,37] reported HPV prevalence by sex or provided data regarding the age of HPV-positive cases, using various age parameters and cut-off values (Table 4). Two [22,27] of these 12 studies also reported the prevalence of HR HPV by sex (footnote *) (Table 4) and 3 [22,28,34] reported prevalences of HPV-16 and HPV-18 by sex. An additional study reported the mean age of patients with HR HPV-positive cancer of the glottis but did not provide estimates of HPV prevalence by sex [37].
Study methodology varied across the included studies (Table 2 and Supplementary Table 3). Six studies were case series [11,22,25,26,28,38], including 1 comparing immunohistochemistry for p16 with a gold standard of polymerase chain reaction (PCR) for the detection of HPV in tumor samples [25]. Six studies were retrospective cohorts designed to evaluate overall or progression-free survival based on clinical and tumor factors, including HPV status [23,24,27,33,34,36]. One study evaluated survival using data prospectively collected from patients with cancer of the oropharynx [21]. Other studies were cross-sectional analyses evaluating HPV prevalence in HNSCCs compared with benign conditions affecting the same organ; these included studies evaluating cancer of the glottis [37], tongue [29], and tonsil [31,35]. Another comparative analysis evaluated HPV prevalence in cancer of the tonsil versus non-tonsillar cancer of the upper aerodigestive tract [32].
Most of the included studies assessed data primarily collected before 2011 [21,23,24,26,28-38], but some assessed data primarily collected from 2011 onwards [11,25,27], including 1 study reporting on patients diagnosed with cancer of the oropharynx from 2016 through 2022 [22]. In total, 13 of the included studies had sample sizes of less than 120 and were conducted in a single hospital [23,24,26,28-34,36-38]. One study based in a dental hospital had a sample size of 187 [27], while 2 other studies conducted across multiple hospitals had sample sizes ranging from 175 to 466 [25,34]. One study did not specify where it was conducted [35].
Among the included studies, HPV genotyping was primarily conducted by DNA chip using commercially available kits (Supplementary Table 3) [24-27,32-34,38]. Other studies conducted HPV genotyping using PCR [22,25,28], cloning [23], and fluorescence scanning [29-31,37]. One study, published in 2004, assessed HPV genotypes using an in-house microarray for HPV DNA [35]. Several studies used p16 detected by immunohistochemistry with a predefined cut-off to test for HPV or HR HPV [11,21-25,31,33,34,36]. However, except for Park, et al. (2012) [24], Jun, et al. (2022) [11], and Lee, et al. (2013) [36], studies did not require p16 expression to define the detection of any HPV or HR HPV.
Prevalence of HPV in patients with head and neck cancer
Multiple studies reporting a range of prevalence values contributed to the calculation of weighted mean prevalence estimates of any HPV and HR HPV for cancers of the glottis [30,37], oropharynx [11,21-25], oral cavity [11,25-28], tongue [11,15,21,27,29,30], and tonsil [11,21,22,25,30-36]. Prevalences of any HPV genotypes and HR HPV for cancer of the gums were based on a single study [27]. HNSCC subtypes with the highest prevalence of any HPV were cancers of the oropharynx (weighted mean, 56.5%; range, 14.5%-75.1%), tonsil (weighted mean, 51.6%; range, 13.9%-80.7%), and posterior wall (28.6%; range, 0.0%-33.3%) (Fig. 2). HNSCC subtypes with the highest mean prevalence of HR HPV were cancers of the tonsil (weighted mean, 48.8%; range, 11.1%-78.9%) and oropharynx (weighted mean, 49.5%; range, 10.8%-72.7%).
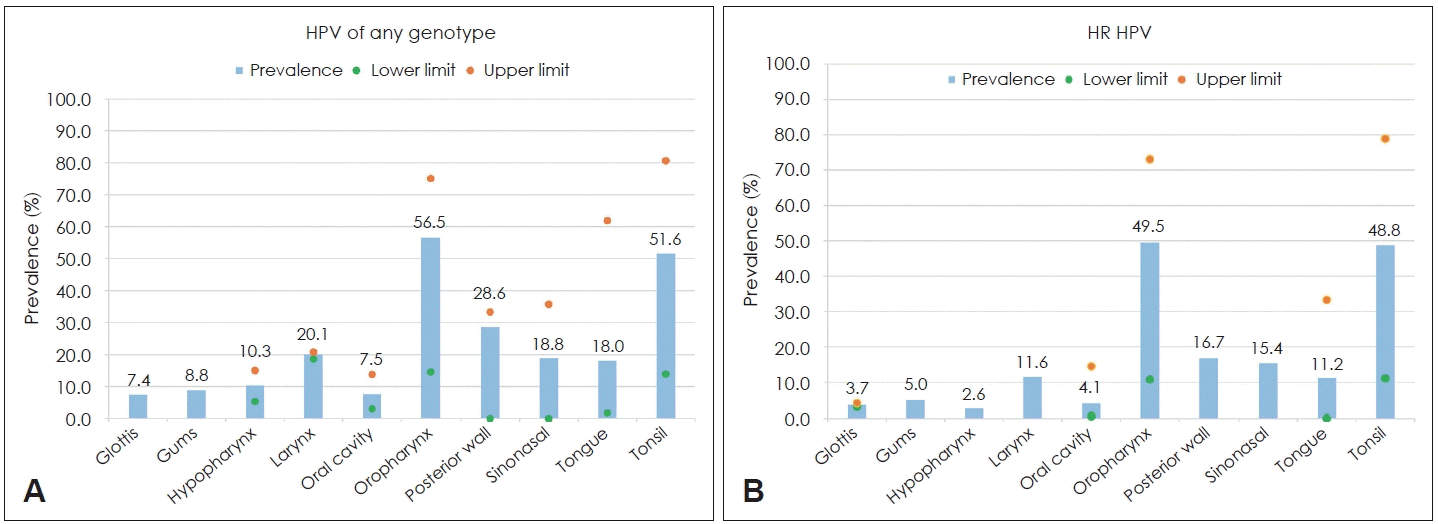
Prevalence of HPV of any genotype and HR genotypes in subtypes of head and neck cancer in the pooled sample of included studies. Bars represent the weighted mean prevalences; weights were determined by the study sample size. Dots represent the lower and upper limits of the range of prevalences. Bars without dots represent estimates derived from a single study. Two studies of cancer of the glottis both reported prevalences of any HPV of 7.4%; thus, a range is not shown. Prevalences cannot be compared across (A) and (B), as not all studies reported on HR HPV. HPV, human papillomavirus; HR, high-risk.
Across subtypes of HNSCC, prevalences of any HPV varied for male cases (range, 8.6%-66.7%) and female cases (range, 4.2%-84.6%) (Table 4). Across both sexes, the lowest prevalences of any HPV were reported for oral cancer [27] and the highest prevalences, for cancer of the oropharynx cancer [21,24,36]. The weighted mean prevalence of any HPV in HNSCCs was higher in male cases (37.9%) than in female cases (24.1%). The mean weighted prevalence of HPV-16 was lower in male cases (7.0%) than in female cases (24.3%), whereas the weighted mean prevalence of HPV-18 was similar across sexes (male: 8.8%; female: 9.6%). Studies varied regarding their definitions of age groups, as well as findings regarding the prevalence of HPV by age.
Genotype distribution of HPV detected in head and neck cancer
The distribution of detected HPV genotypes varied widely by study and HNSCC subtype (Fig. 3). HPV-16 was the most common genotype detected. The weighted mean proportion of HPV-positive samples in which HPV-16 was detected was 75.6% for cancer of the oropharynx, 74.1% for cancer of the tonsil, 50.0% for cancer of the hypopharynx, 50.0% for cancer of the glottis, 40.0% for sinonasal cancer, 37.5% for cancer of the larynx, 29.5% for tongue cancer, and 26.8% for oral cavity cancer. HPV-18, the second-most common genotype detected, accounted for an average of 11.8% of HPV-positive oral cavity cancers, 9.0% of HPV-positive cancers of the oropharynx, and 8.6% of HPV-positive cancers of the tonsil. The weighted mean proportion of HPV infections involving HPV-33 was 2.3% for cancer of the oropharynx, 2.0% for cancer of the tonsil, and 1.9% for oral cavity cancer. HPV-35 was detected in an average of 7.2% of HPV-positive cancers of the glottis, 2.7% of HPV-positive cancers of the tonsil, and 2.6% of HPV-positive cancers of the oropharynx. HPV-52 was detected in an average of 16.7% of HPV-positive sinonasal cancers. HPV-58 was detected in an average of 1.9% of HPV-positive cancers of the oropharynx and 1.3% of HPV-positive cancers of the tonsil. HPV-69 was detected in an average of 20.0% of HPV-positive sinonasal cancers and 0.6% of HPV-positive cancers of the tonsil.
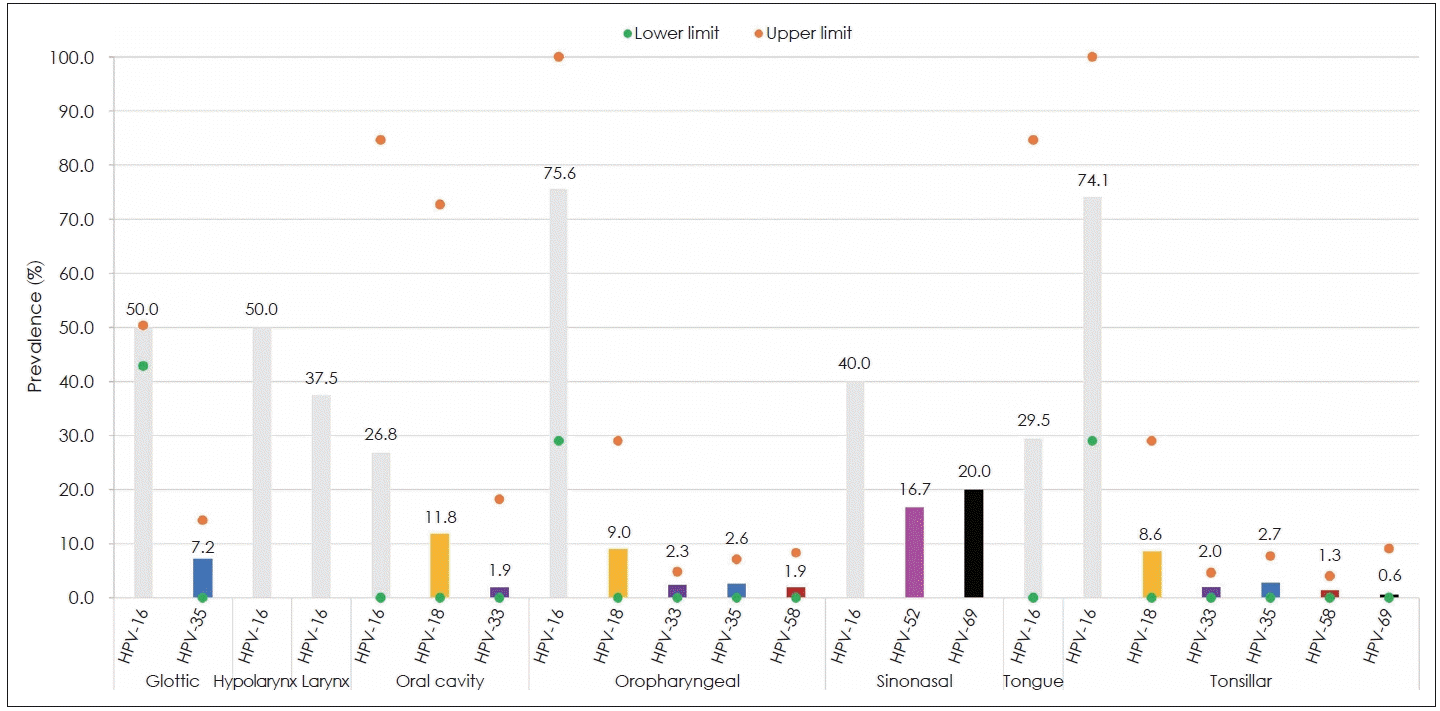
Distribution of HPV genotypes in subtypes of head and neck cancer, estimated from the pooled sample of included studies. Bars represent the weighted mean of the proportion HPV-positive cases in which the genotype was detected; weights were determined by the study sample size. Dots represent the lower and upper limits of the range of proportion detected. Bars without dots represent estimates derived from a single study. Proportions were estimated for each detected HPV genotype, regardless of whether it was part of a co-infection. HPV, human papillomavirus.
Discussion
The current review evaluated studies reporting on the prevalence and genotype distribution of HPV among South Korean patients diagnosed with HNSCC, finding that HPV was detected in a substantial number of cancers, including 56.5% of cancers of the oropharynx, 20.1% of cancers of the larynx, and 7.5% of oral cavity cancers. This finding fills a significant knowledge gap, as data on South Korea have been underrepresented in previous reviews of HPV-positivity in HNSCCs. While several reviews have estimated HPV prevalence across HNSCC subtypes using data from 30 to 273 studies globally and 4 to over 60 studies for East Asia [7,39-42], these estimates often include a diverse and inconsistently defined mix of Asian countries. Moreover, only 1 to 4 studies specifically focused on South Korean patients have been included in these reviews, highlighting the need for more region-specific data to better understand the burden of HPV-related HNSCC in South Korea [5,7,39-42].
Despite the limitations of previous reviews, we noted that our findings on the prevalence of HPV in HNSCC broadly accord with similar, albeit widely varying, estimates previously reported for Asia, which ranged from 20.4% to 51.7% for cancer of the oropharynx, 1.0% to 42.8% for oral cavity cancer, and 1.4% to 49.1% for cancer of the larynx [7,8,39-42]. Data from the current review indicate that in South Korea HR HPV accounts for 70% to over 90% of HPV genotypes detected in cancers of the oropharynx, tongue, tonsil, and oral cavity. The HR HPV-16 genotype was particularly common among cases in our data, representing about three-quarters of all genotypes detected in cancers of the oropharynx and tonsil and over one-quarter of genotypes detected in cancers of the larynx and oral cavity. Other reviews of HNSCC globally and in Asia have similarly reported that HPV-16 was overwhelmingly the most common genotype detected in HNSCC, particularly in cancer of the oropharynx, where it represented 75.9% to 97.8% of detected genotypes [7,40,42]. Although some co-infections were reported in the 19 studies included in this review, all involved HPV-16 or other HR genotypes, a finding consistent with reports of co-infection in previous reviews of HPV-associated HNSCC [39,42]. Together, these findings align with the understanding that oral infection with HR HPV genotypes is an important cause of HNSCCs in South Korea and globally.
We observed that the weighted mean average prevalence of any HPV was higher in male than female cases of HNSCC in South Korea. Previous studies have reported that compared with females, males have a 2- to 3-fold higher prevalence of HPV-16 in cancer of the oropharynx [43] and account for 2 to 5 times as many cases of HPV-associated HNSCCs [5]. The male preponderance of HPV in HNSCCs may, in part, be explained by biological factors, including the greater transmissibility of oral HPV to males compared to females during oral sex [44] and cross-protective antibody responses from HPV cervical infections in females [45,46]. HPV vaccination of males may help reduce the burden of HNSCCs in South Korea. The nonavalent vaccine, available in South Korea, targets HPV genotypes, 6, 11, 16, 18, 31, 33, 45, 52, and 58. Notably, HPV-33 and HPV-58 were detected in a substantial number of HNSCC cases in the studies we reviewed.
Extensive between-study variability in the prevalence of any HPV and HR HPV in HNSCC tumors was observed in this review, a finding consistent with those of other reviews of HNSCC that have reported extensive heterogeneity in HPV prevalence within a single region or country [39,40,42]. Extensive variability is also apparent in cross-regional comparisons of HPV prevalence, with some reviews reporting a higher prevalence of any HPV in cancer of the oropharynx in Asia than other world regions (46.3% vs. 35.6% [40]), some a similar prevalence (20.4% vs. 24.9% [7]), and some a lower prevalence (36.4% vs. 48.5% [41]). In some countries, including the United States, Australia, Canada, Japan, and Singapore, the incidence of HPV-positive cancer of the oropharynx has remained steady or increased in recent decades, while that of HPV-negative cancers of the oropharynx has decreased, possibly due to declines in tobacco and alcohol use [47-49]. In contrast, several countries in Europe have reported concomitant increases in both HPV-positive and HPV-negative cancers of the oropharynx [49]. Notably, varying incidence trends in HPV-positive HNSCC may reflect both geographic and temporal variations in sexual risk factors for oral HPV [37,50-53], as well as changes in oral HPV prevalence in the general population [54]. Given the variable temporal trends in HPV-positive HNSCC, cross-study differences in HPV prevalence in the current review may in part be due to differences in the years of case selection, which ranged from 1991 to 2022.
As reported in other reviews [39-42], a range of HPV detection and genotyping methodologies were used in studies evaluating HNSCC in South Korea, which may have further contributed to cross-study variation in prevalence estimates. Although most of the included studies used PCR for HPV detection, the sensitivity and specificity of PCR-based assays can vary widely [55]. Among studies in the current review, PCR techniques, though not always fully described, varied with regard to being endpoint-based versus a quantitative assay, the sequence target (L1 vs. L1/E6/E7), and the use of a commercial DNA chip versus other approaches. Notably, PCR may detect transient HPV infections unrelated to the malignancy, potentially leading to an overestimation of the burden of HPV-related tumors [46]. To address this issue, studies of HPV positivity in HNSCC tumors may evaluate other cellular markers to validate HPV-related oncogenesis [46]. Notably, p16 overexpression, an indicator of post-translation dysregulation detectable by immunohistochemistry, is recommended as a surrogate indicator of HR HPV and for use in tumor staging for oropharyngeal squamous cell carcinoma [46,55]. Another cellular marker, viral E6/E7 mRNA expression, indicates transcription of key HPV oncoproteins, correlates highly with p16 immunohistochemistry findings and is considered the gold standard of HPV-positivity in oropharyngeal tumors [49,55]. In the current review, most studies evaluating cancers of the oropharynx reported immunohistochemistry results for p16, most frequently [15,21,22,24,31,33,34,36], but not always [11,23] using a threshold of greater than 70%. Several studies also evaluated oncogenesis markers, including E6/E7 viral mRNA, E2/E6 viral mRNA, PD-L1, EGRF, pRB, and c-MET [21,29-31,33,37].
The current review had some limitations, which should be considered when evaluating its findings. Definitions of the anatomical sites varied across the included studies and were not always clearly described. As this was a systematic literature review, our findings depend on how the included reference studies reported their data. For example, if the studies did not separate oropharyngeal cancer subtypes, we could not disaggregate and identify the specific components (e.g., tonsil, soft palate, tongue) they collected. In addition, some of the studies were not conducted by head and neck specialists, which may have led to inaccuracies in classifying the anatomical sites. Furthermore, in some studies, cancer of the tonsil was reported as a separate category, potentially leading to a double counting of cases, as the tonsils are part of the oropharynx. Double-counting may also have occurred in studies reporting separately for tongue cancer, which is a subtype of oral cavity cancer and oropharyngeal cancer. Case selection was not well described in several studies, and thus the generalizability of their findings is unclear. Some studies assessed only a few genotypes of HPV, which may have led to an underestimation of HPV prevalence. The number of HPV genotypes tested varied across included studies, ranging from 3 to 42, potentially affecting findings regarding the distribution of HPV genotypes in HNSCC tumors. Additionally, the assays used for HPV genotyping in different studies varied in the number of HPV genotypes detected, which may have contributed to inconsistencies in genotype detection across studies. Finally, 3 studies lacked data on genotype or HR HPV status and thus were excluded from HR HPV prevalence estimates, potentially affecting the accuracy of these estimates [11,21,36].
In conclusion, we found that HR HPV, particularly HPV-16, was commonly detected in both male and female patients with subtypes of HNSCC in South Korea, particularly in cancer of the oropharynx. Safe and effective HPV vaccines are currently in use among girls only in South Korea [14,15,56]. To decrease the future burden of HPV-associated HNSCCs in South Korea, consideration should be given to extending the use of HPV vaccines to males and boys.
Supplementary Materials
The Supplement is available with this article at https://doi.org/10.3342/kjorl-hns.2025.00276.
Notes
Acknowledgments
The authors thank Ingrid Peterson in collaboration with ScribCo for medical writing assistance.
This study was funded by Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA.
AFSM was an intern of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA at the time of the study.
RW and WW are employees of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA and may have stock options in Merck & Co., Inc., Rahway, NJ, USA.
SO and GSY are employees of MSD Korea and may have stock options in Merck & Co., Inc., Rahway, NJ, USA.
YHW is an employee of MSD Taiwan.
IS is an employee of MSD Thailand and may have stock options in Merck & Co., Inc., Rahway, NJ, USA.
SYL has no conflicts of intertest to report.
Author Contribution
Conceptualization: Aneesha Fathima Syed Mohamed, Ying Hui Wu, Wei (Vivian) Wang. Data curation: Aneesha Fathima Syed Mohamed, Wei (Vivian) Wang. Formal analysis: Aneesha Fathima Syed Mohamed, Seyoung Oh, Ying Hui Wu, Ying Hui Wu, Isaya Sukarom, Sei Young Lee, Wei (Vivian) Wang. Funding acquisition: Wei (Vivian) Wang. Investigation: Seyoung Oh, Ying Hui Wu, Ying Hui Wu, Isaya Sukarom, Sei Young Lee, Wei (Vivian) Wang. Methodology: Aneesha Fathima Syed Mohamed, Seyoung Oh, Ying Hui Wu, Ying Hui Wu, Isaya Sukarom, Sei Young Lee, Wei (Vivian) Wang. Project administration: Aneesha Fathima Syed Mohamed, Ruixuan Wang. Resources: Aneesha Fathima Syed Mohamed, Ruixuan Wang, Wei (Vivian) Wang. Software: Aneesha Fathima Syed Mohamed. Supervision: Aneesha Fathima Syed Mohamed, Ruixuan Wang, Ying Hui Wu, Wei (Vivian) Wang. Validation: Aneesha Fathima Syed Mohamed. Visualization: Aneesha Fathima Syed Mohamed. Writing—original draft: all authors. Writing—review & editing: all authors.